Genetic analysis has potential to transform diagnosis and treatment of adults with liver disease of unknown cause
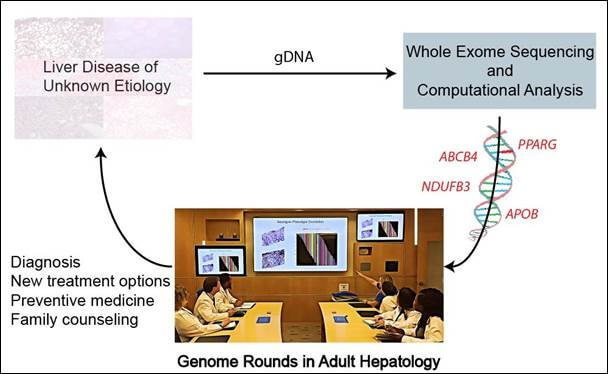
Whole-exome sequencing successfully diagnosed genetic cause of liver disease in one in four adults and could play a key role in timely diagnosis and treatment of adults with liver disease of unknown origin, report scientists in the Journal of Hepatology
Amsterdam, April 15, 2019 – Adults suffering from liver disease of unknown cause represent an understudied and underserved patient population. A new study reported in the Journal of Hepatology, published by Elsevier, supports the incorporation of whole-exome sequencing (WES) in the diagnosis and management of adults suffering from unexplained liver disease and underscores its value in developing an understanding of which liver phenotypes of unknown cause in adults are genetic.
Chronic liver disease is a significant health problem affecting more than four million people in the United States and leading to over 40,000 deaths annually. It often remains undiagnosed for many years unless there is awareness of subtle clinical signs, behavioral risk factors, and/or investigation of abnormal liver function tests. In many patients, by the time overt manifestations of chronic liver disease emerge, liver injury has already advanced resulting in portal hypertension or hepatic decompensation.
WES, which consists of sequencing all the approximately 20,000 human protein-coding genes, is attractive and suitable for clinical use and translational research studies. “Advances in human genomics through next generation sequencing technology have created an unprecedented opportunity for genetic investigation and clinical diagnosis,” explained lead investigator Silvia Vilarinho, MD, PhD, Assistant Professor of Medicine (Digestive Diseases) and Pathology, Departments of Internal Medicine and Pathology, Yale School of Medicine, New Haven, CT, USA. “However, to date, most studies that investigate the use of next generation sequencing technologies in diagnosis and personalized medical care have been performed in either pediatric or cancer patients. The clinical utility of these approaches for a broader spectrum of diseases among adults remains poorly studied.”
Investigators assessed the utility of WES in diagnosis and management of adults with unexplained liver disease despite comprehensive evaluation by a hepatologist and with no history of alcohol overuse. They performed WES and deep phenotyping of 19 unrelated adult patients with idiopathic liver disease recruited at a tertiary academic healthcare center.
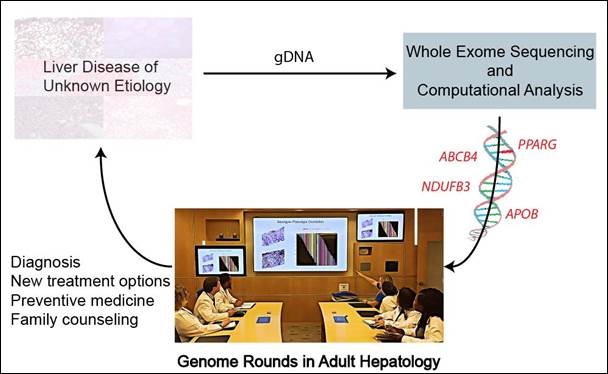
Analysis of the exome in 19 cases identified four monogenic disorders in five unrelated adults, enabling new treatment. This strategy also shed further light on genetic contributions, and therefore underlying molecular pathogenesis, across different forms of liver disease that are clinically indistinguishable through conventional diagnostic approaches.
“This study provides evidence that a subset of adult patients who suffer from liver disease of indeterminate etiology with or without other comorbidities harbor an underlying Mendelian disorder, which may be unrecognized during their entire childhood until genetic testing is performed,” commented Dr. Vilarinho. “Our data highlight the importance of using WES in the investigation of liver disease of unknown cause so that we may start developing an understanding of what clinical presentations or diseases are genetic and may remain undiagnosed until adulthood.”
“WES testing is transforming the medical practice of undiagnosed diseases,” commented Filippo Pinto e Vairo, MD, PhD, Center for Individualized Medicine and Department of Clinical Genomics, and Konstantinos N. Lazaridis, MD, Center for Individualized Medicine and Division of Gastroenterology and Hepatology, both at the Mayo Clinic, Rochester, MN, USA, in an accompanying editorial. “Of note, the current cost of clinical grade WES testing and interpretation is comparable to some advanced imaging exams (such as MRI) or endoscopic procedures that we already use in practice.”
“Based on the experience of Dr. Vilarinho and colleagues, and our own work within the Mayo Clinic Center for Individualized Medicine, we strongly advocate the use of WES testing in the clinic for adult patients with idiopathic liver disease, who have onset of symptoms at up to 40 years of age. Moreover, discoveries from such testing could also assist the individualized management and follow-up of these patients,” concluded Dr Lazaridis.
—
Notes for editors
The articles are “Clinical utility of genomic analysis in adults with idiopathic liver disease,” by Aaron Hakim; Xuchen Zhang; Angela DeLisle; Elif A Oral; Daniel Dykas; Kaela Drzewiecki; David Assis; Marina Silveira; Jennifer Batisti; Dhanpat Jain; Allen Bale; Pramod K. Mistry; and Silvia Vilarinho (http://dx.doi.org/10.1016/j.jhep.2019.01.036) and “Individualized medicine comes to the liver clinic,” by Filippo Pinto e Vairo and Konstantinos N. Lazaridis (https://doi.org/10.1016/j.jhep.2019.03.025). They will appear in the Journal of Hepatology, volume 70, issue 6 (June 2019) published by Elsevier.
Full text of these articles is available to credentialed journalists upon request; contact Carolien van der Zanden at +31 20 485 3346 or hmsmedia@elsevier.com. Journalists wishing to interview the study authors should contact Dr. Silvia Vilarinho, at silvia.vilarinho@yale.edu. Dr. Lazaridis may be reached for comment at lazaridis.konstantinos@mayo.edu.
About the Journal of Hepatology
The Journal of Hepatology is the official journal of the European Association for the Study of the Liver (EASL). It publishes original papers, reviews, case reports, and letters to the Editor concerned with clinical and basic research in the field of hepatology. www.journal-of-hepatology.eu
About EASL
In the fifty plus years since EASL was founded, it has grown from a small organization that played host to 70 participants at its first meeting, to becoming the leading liver association in Europe. EASL attracts the foremost hepatology experts as members and has an impressive track record in promoting research in liver disease, supporting wider education, and promoting changes in European liver policy. www.easl.eu
About Elsevier
Elsevier is a global information analytics business that helps scientists and clinicians to find new answers, reshape human knowledge, and tackle the most urgent human crises. For 140 years, we have partnered with the research world to curate and verify scientific knowledge. Today, we’re committed to bringing that rigor to a new generation of platforms. Elsevier provides digital solutions and tools in the areas of strategic research management, R&D performance, clinical decision support, and professional education; including ScienceDirect, Scopus, SciVal, ClinicalKey and Sherpath. Elsevier publishes over 2,500 digitized journals, including The Lancet and Cell, 39,000 e-book titles and many iconic reference works, including Gray’s Anatomy. Elsevier is part of RELX Group, a global provider of information and analytics for professionals and business customers across industries. www.elsevier.com

